 .jpg)
When one of these neutrons collides with a nitrogen - 14, a carbon - 14 atom and a proton, 1H result.ĥ. Write the decay equation for an excited atom of Cobalt-60 undergoing gamma decay. First, we need to convert the 1.00 g of sample into number of atoms of cobalt-60 and to convert the activity into numbers of atoms that decay per second. Write nuclear equation for the formation of cobalt-60 Express your answer as a nuclear equation Submit My. It is produced by irradiation of cobalt-59 in a nuclear reactor. (2) In Earth's upper atmosphere, energetic cosmic rays cause atmospheric gas atoms to emit neutrons. Problem 11.51 Part A Cobalt-60 (half-life 5.3 years) is used to irradiate food, to treat cancer, and to disinfect surgical equipment. The list of radionuclides excludes those with half lives measured in seconds. For example, cobalt-60 undergoes beta-minus. 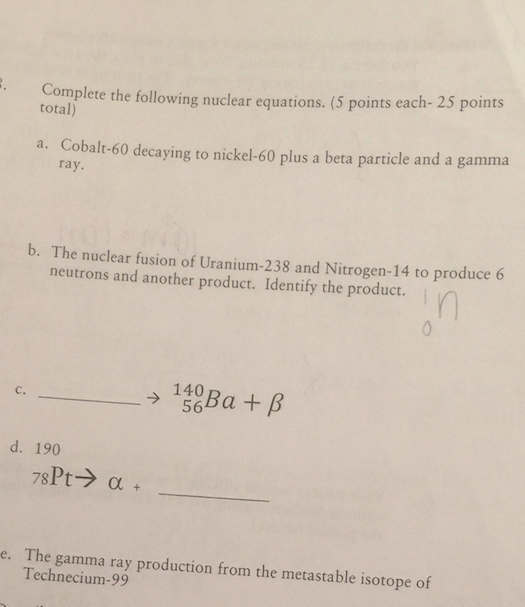
(1) K-40, a beta particle emitter, is a naturally occurring potassium isotope found in some foods. This Web application will allow you to calculate the activity of a radionuclide after a specified interval of time. Unlike other types of radioactive decay, gamma decay does not change the nuclear composition of the nuclide. N = No(exp)-kt where N is the number of counts and t is time.įind a simple expression for the half life.Ĥ.(1pt) Write the appropriate nuclear equation for each of the following descriptions. If 4.71g of this nuclide register 20 counts per minute on a radioactivity counter, what is the half life of the nuclide? What percentage of Cobalt atoms are Cobalt-60?Ģ.(2pts) A radioactive nuclide of mass number 94 has been prepared by neutron bombardment. Over time cobalt-59 absorbs a neutron to become cobalt-60. RADIOISOTOPE BRIEF Cobalt-60 (Co-60) Half-life ( 5. Placing this non-radioactive, Cobalt-59 pellets into a nuclear reactor creates deliberately produced cobalt-60. How much of a 1.000 mg sample will remain after 15.9 years. By bombarding cobalt 59 with neutrons, in a nuclear reactor, an additional neutron can be captured by the nucleus converting it into cobalt 60. These sorts of cascades are the bread and butter of nuclear physics the double-photon decay paper you found is much rarer.1.(2pts) A radioactivity counter gives a reading of 350 cts per min for a 12.5 mg sample of Cobalt (II) Chloride partially enriched with Cobalt-60 (t1/2 = 5.26 yr.). Example: The half-life of cobalt-60 is 5.3 years. That photon must carry lots of orbital angular momentum, in addition to its spin, so the first excited state of cobalt is a relatively long-lived isomer (about ten minutes).Ĭobalt-60 is used as a gamma source because it decays to an excited state of the nickel-60 nucleus, which then cools by emitting a sequence, or a "cascade," of photons. Since cobalt-60 has ground state spin-parity $5^+$, first excited state $2^+$, a single photon can mediate the transition. + 4He 27 0 2 Solution mass numbers 60 60 59Co + 1n 56Mn + 4H e 27 0 25 2 27. Usually, in nuclear decays, magnetic-dipole transitions are suppressed compared to electric-dipole transitions. 
The paper you have linked measures a rare mode where two real photons are produced, and a surprising observation that in the double decay $E$-type photons are produced at the same rate as $M$-type photons. (See Table VIII for a list of items that are sterilized.) Increased use for food irradiation is likely in the future. 
Most of its current use is in the sterilization industry, primarily for medical products intended for human consumption. Mostly they decay by emitting a "virtual" photon, which produces a real positron-electron pair in the field of the nucleus. Cobalt-60 (half-life 5.27 years) is the largest revenue-producing commercial radioisotope in the world. Since a single photon must carry away at least one unit of spin, these excitations cannot decay by one-photon emission. Both of these nuclides have a first excited state with spin-parity $0^+$, the same as their ground state. Complete the nuclear equation in your answer book. Explain in terms of BOTH protons and neutrons, why Co-59 and Co-60 are isotopes of cobalt. Small nickel-plated slugs of the radioactive metal are loaded into a sealed alloy cylinder typically 10 × 450 mm and doubly encapsulated in a corrosion-resistant steel pencil. The incomplete equation for the decay of cobalt-60, including beta and gamma emissions, is shown below. The paper that you cite describes decays in calcium-40 and zirconium-90 by emission of two photons at once. Cobalt-60 is produced by neutron bombardment of stable cobalt in a nuclear reactor. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
